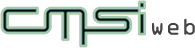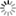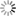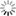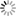Mechanism of hydrogen and methane hydrate formation and dissolution and thermodynamic stability
Interviewee:
|
Interviewer:
|
| Professor, Graduate School of Natural Science and Technology, Okayama University |
CMSI Molecular Science |
What are hydrogen hydrate and methane hydrate?
Hydrates are crystalline solids consisting of water molecules and other gaseous molecules formed in high-pressure environments. In hydrogen hydrates, hydrogen molecules are encapsulated in water lattice. Depending on the temperature and pressure, hydrogen hydrate may form a cage-like clathrate hydrate structure, or the hydrogen may be contained inside the interstitials in ordinary ice. The occupancy of hydrogen in the ice will also vary depending on the conditions. The term "methane hydrate" is often heard on TV these days. This is a methane clathrate hydrate, with a structure in which methane is contained inside a cage made of water molecules . In the real world, the cage-like structure is not formed in the absence of methane.
How much progress has there been in research in this area?
We have studied hydrates for around 20 years, so some knowledge has been accumulated in this area. Even a less computationally demanding method enables to obtain a phase diagram (figure below) of the structure obtained, which is in considerable agreement with the experimental results. If pressure is applied to hydrogen hydrate, it is possible to trap hydrogen in ice up to a mass of about one-ninth of water. However, this requires a pressure of 20,000 atmospheres, making it absolutely impossible to load the substance on a vehicle and transport it. However, other substances can be added to reduce the pressure, so this process is expected to find applications.
Currently, large-scale simulations for dissociation of methane hydrate are being conducted on the K computer using the molecular dynamics software program Modylas. The simulations show that when methane hydrate is heated, it melts from the surface inward, but it takes a long time when it is surrounded by water. When methane is placed in liquid water, this results in oversaturation due to its low solubility and any further dissolution of methane is prevented. At higher concentrations, however, bubbles are formed, and the methane escapes into these bubbles, so the hydrate once again begins to melt. Simulations are enabling us to begin to understand processes like these.
Why is research being conducted into how methane hydrate melts?
It is known that there is twice the reserve of methane hydrate as other fossil fuels, and it is expected in industrial usage. However, methane hydrate is found on the ocean floor, and at least 87% of its mass is composed of water, so excavating it in solid form would not be cost-effective. We need to find a way of melting it prior to excavation and gathering together just the gaseous methane. In thermodynamic terms, it is possible to melt the methane by burning it with the quantity of heat required to burn about 10-20% of the total quantity of methane, but various conditions are needed for it. Simulations are being performed to study these conditions.
What are your plans for future research in this area?
For hydrogen hydrate, we are working to develop models and theories in order to improve accuracy in predicting its thermodynamic stability. For methane hydrate, we plan to conduct even larger-scale simulations in order to examine dissociation kinetics by performing simulations in the presence and absence of methane bubbles. Current simulations do not allow us to track formation and growth of bubbles in water.
We are also making plans to use simulations to determine whether it is possible to melt the hydrate by applying external field.
|
|
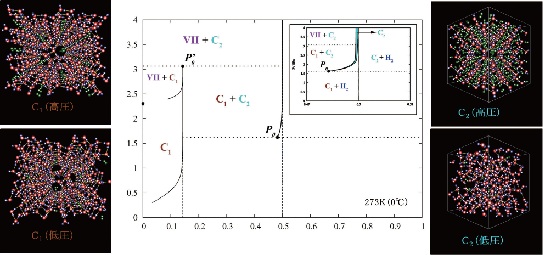
| Phase diagram of hydrogen hydrate derived from simulations: This figure shows the types of hydrogen hydrates that are produced at 0 °C with various pressures and hydrogen compositions . C₁ and C₂ are types of hydrogen hydrate. C₁ has hydrogen encaged in a diamond-like cubic crystal ice known as Ice Ic, while C₂ has hydrogen encaged in an orthorhombic crystal ice known as Ice II. VII is cubic crystal ice with a different structure. H₂ indicates a state in which the hydrogen is not encaged in ice but exists outside in liquid form. Regions denoted by two labels stand for coexistence of the corresponding two phases. The images on the left and right show the structures of C₁ and C₂, respectively. In the images, green hydrogen molecules are encaged in the lattice of red and blue water molecules. Under high pressure, there are large numbers of hydrogen molecules encapsulated in each cage , but under low pressure there are only a few. In the case of C₂, the ice crystal structure breaks down at low pressure. |